Drugs
01
AL101-AOB
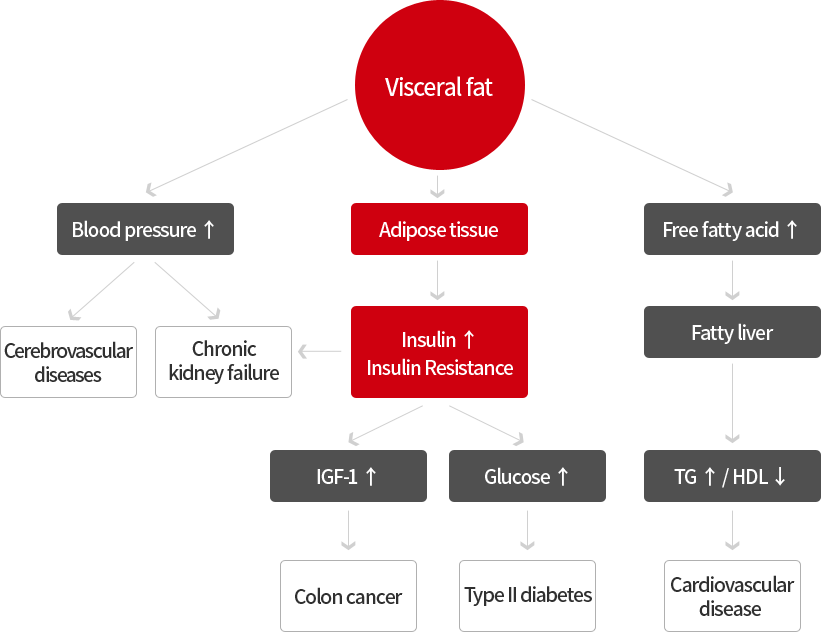
Obesity has emerged as a major public health problem due to its high prevalence and association with increased risk towards the development of a metabolic syndrome
The incidence of lifestyle diseases like hypertension, diabetes, hyperlipidemia, various heart diseases and stroke etc. is much higher for people who have a visceral type of abdominal fatness, even if they have the same Body Mass Index (BMI) like others
Subcutaneous fat and visceral fat are metabolically different
Visceral fat contributes to the pathogenesis of the underlying metabolic syndrome

AL101-AOB
Background of |
|
|---|---|
Summary of |
|
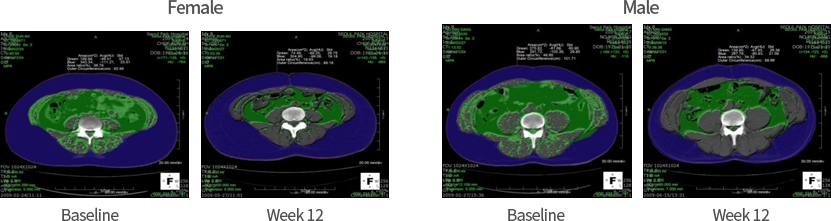
Reduction of visceral fat by 15% after a 12-week treatment of AL101-AOB (CT scan)

02
AL101-AMD
Background of |
|
|---|---|
Development Objective |
|
Summary of |
|
Competitors
Classification |
Lucentis |
Eylea |
Vabysmo |
AL101-AMD |
|---|---|---|---|---|
Company |
Genentech |
Regeneron |
Roche |
AngioLab |
Active ingredient |
Ranibizumab |
Aflibercept |
Faricimab-svoa |
ALS-L1023 |
Mechanism |
Anti-VEGF antibody fragment |
Fusion Protein of VEGFR |
VEGF-A and Angiopoietin |
VEGF, bFGF, PDGF, MMPs inhibition |
Side effect |
Increased risk of stroke, |
Increased risk of stroke, |
Conjunctival hemorrhage, |
No adverse effects |
Remarks |
FDA Approved(2006) |
FDA Approved(2011) |
FDA Approved(2022) |
Orally administered |
03
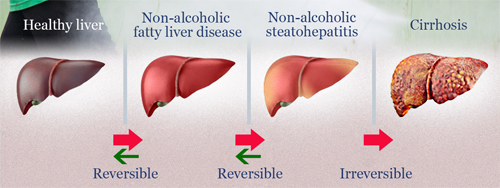
AL101-MASH
Background of |
|
|---|---|
Development Objective |
|
Summary of |
|
04
AL102-PDT
Background of |
|
|---|---|
Development Objective |

|
Summary of |
|
05
AL101-OME
Background of |
|
|---|---|
Development Objective |
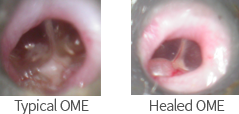
|
Summary of Product |
|
06
AL101-ROSA
Background of |
|
|---|---|
Development Objective |
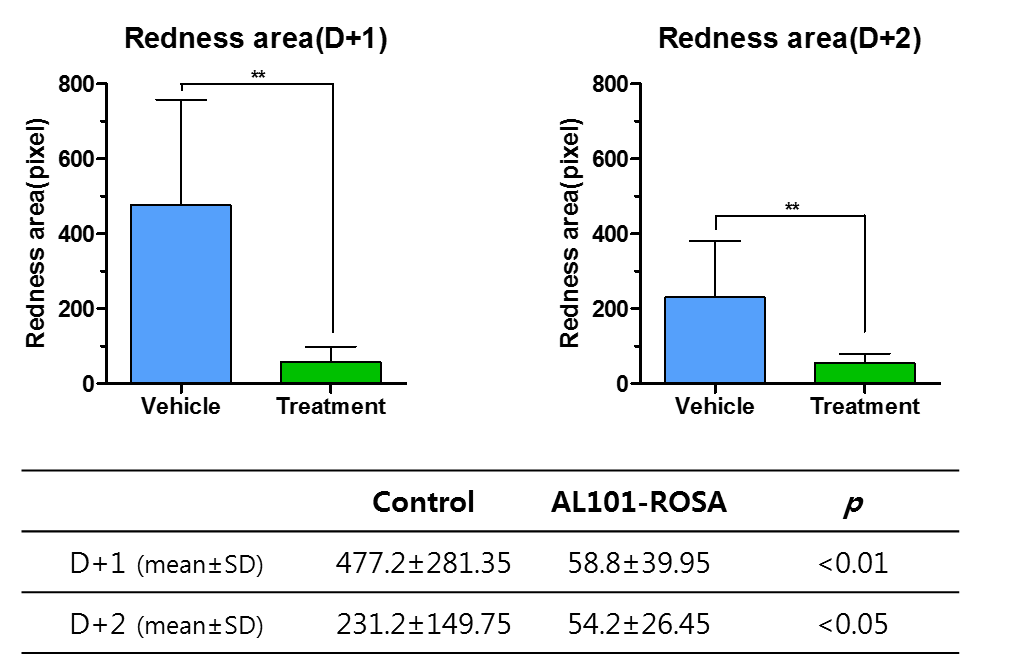
|
Summary of Product |
|
07
AL101-PSO
Background of |
|
|---|---|
Development Objective |
|
Summary of Product |
|
08
AL201-AB
Background of |
|
|---|---|
Development Objective |
|
Summary of Product |
|
